Introduction
Our research is integrative of two fields of neural plasticity: the neurobiology of learning and memory and neural plasticity after brain damage, including stroke. Neural plasticity is the mechanism underlying the capacity to encode new experiences and change behavior throughout the lifespan, i.e., to learn. Neural plasticity is also instigated by brain damage in the form of regenerative responses, in which surviving neurons attempt to restore more normal brain function by growing and forming new connections with one another. Neural regenerative responses appear to be intrinsically sensitive to behavioral experiences. As a result, behavioral changes that occur soon after brain damage can interact with regenerative responses to influence the pattern of brain reorganization. This is relevant because brain damage survival often demands profound behavioral changes to compensate for impairments. Stroke survivors with impairments in one hand learn new ways of relying on the better functioning hand. This has two major implications: (1) Understanding how the brain normally responds to an injury requires understanding how this response is shaped by behavioral changes that normally follow that injury. (2) It should be possible to shape brain reorganization in a manner that optimizes functional outcome with manipulations of behavioral experiences. Clearer understanding of each of these could be central to endeavors to promote adaptive brain plasticity and improve function after brain damage. Our ongoing efforts to clarify them are overviewed below.
How is brain reorganization shaped by behavioral compensation?
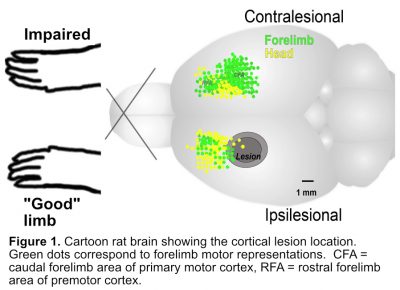
Strokes often result in chronic impairments in one hand, resulting in compensatory reliance on the other hand. These impairments can be modeled in rodents with stroke-like damage to the region of cortex that mediates forelimb sensation and movements (Fig. 1). Like humans, rats compensate for loss of function in one forelimb by relying more on the better functioning (“good”) limb.
In early work to determine whether behavioral compensation influences brain remodeling responses, we examined the motor cortex in the hemisphere opposite to the damaged region (“contralesional” motor crotex). This was an obvious place to look because it mediates function of the good forelimb and undergoes regenerative reactions triggered by the degeneration of neural connections with the damaged region. Indeed, we found that this region undergoes a sequence of cellular and structural changes including astrocytic reactivity, increased expression of growth factors, dendritic growth, synaptogenesis and alterations in the configuration of synaptic connections, as detected using various sensitive quantitative methods for light- and electron-microscopy (Fig. 2). Experimental manipulations revealed that the dendritic and synaptic growth responses were dependent upon increased reliance on the good forelimb, but only in part. They were also dependent upon the degeneration of neural connections, which instigates regenerative reactions. That is, the neural remodeling responses in contralesional cortex were the result of an interaction between increased reliance on the good limb and regeneration reactions (Fig. 3). Additional studies supported that the capacity to learn a new skill with the good limb was facilitated after motor cortical lesions as a result of its convergence with regenerative responses in the contralesional motor cortex.
 Compensation with the good forelimb can be considered an adaptive response to unilateral brain damage because it helps animals to return to daily activities that enable survival. However, we discovered that skill acquisition with the good limb can come with costs. Even a brief period of training the good forelimb in a novel skilled reaching task, to model the effects of learning new ways of using this limb, worsened function of the impaired forelimb and limited the benefits of rehabilitative motor training focused on the impaired limb after motor cortical lesions in rats. The latter effect was linked with diminished recovery of forelimb movement territory (“motor map”) and an altered pattern of synaptic structural change in remaining motor cortex of the injured hemisphere (Figure 4). The results suggest that learning with the good limb drove synaptic changes in the remaining motor cortex that competed with those that could otherwise be driven by training focused on the impaired limb.
Compensation with the good forelimb can be considered an adaptive response to unilateral brain damage because it helps animals to return to daily activities that enable survival. However, we discovered that skill acquisition with the good limb can come with costs. Even a brief period of training the good forelimb in a novel skilled reaching task, to model the effects of learning new ways of using this limb, worsened function of the impaired forelimb and limited the benefits of rehabilitative motor training focused on the impaired limb after motor cortical lesions in rats. The latter effect was linked with diminished recovery of forelimb movement territory (“motor map”) and an altered pattern of synaptic structural change in remaining motor cortex of the injured hemisphere (Figure 4). The results suggest that learning with the good limb drove synaptic changes in the remaining motor cortex that competed with those that could otherwise be driven by training focused on the impaired limb. 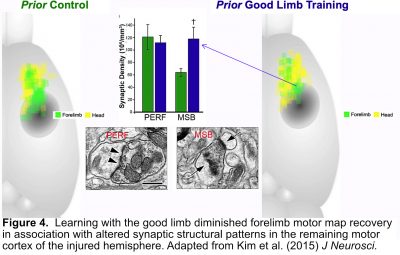
It has long been thought that disuse of the impaired hand after stroke exacerbates impairment by limiting the practice that could support its functional improvements. Our findings suggest that it is not just disuse, but also learning new ways of using the good hand on its own, that can exacerbate impairment. Understanding the neural mechanisms of this phenomenon, how the deleterious consequences of compensation can be avoided, and how the results generalize across different stroke loci are major ongoing research areas. These findings also further spurred our interest in better understanding how behavioral interventions can be used to shape brain reorganization in a manner that optimizes outcome.
Harnessing the Experience-Dependency of Brain Remodeling to Improve Outcome
Manipulations of behavioral experience are likely to be central to endeavors to improve function after brain injury because they capitalize upon the way brains have evolved to reorganize functional connectivity, via learning. Our research in this direction has focused on 2 general topics: (1) neural and behavioral changes induced by motor rehabilitation after cortical injury and (2) the efficacy of combining rehabilitative training with other therapies, including cortical stimulation.
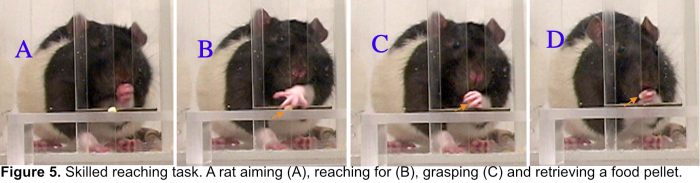
 Others have previously discovered that training the impaired limb in unilateral skilled reach tasks (Fig. 5), to model effects of motor rehabilitative training, promotes recovery and reorganization of remaining forelimb movement territory in motor cortex after motor cortical lesions. We found that this training improves forelimb function in association with increases in a mature efficacious population of synapses in the reorganized region of motor cortex (Fig. 6).
Others have previously discovered that training the impaired limb in unilateral skilled reach tasks (Fig. 5), to model effects of motor rehabilitative training, promotes recovery and reorganization of remaining forelimb movement territory in motor cortex after motor cortical lesions. We found that this training improves forelimb function in association with increases in a mature efficacious population of synapses in the reorganized region of motor cortex (Fig. 6).
Most mechanistic studies of rehabilitative training effects, including our own, have focused on the brain changes that can be observed by the end of the training. The process of change, and its interactions with neural regenerative responses, is minimally understood. To efficiently advance understanding on this topic, we are applying live-animal imaging approaches in collaboration with Dr. Andy Dunn at UT and Dr. Yi Zuo at University of California Santa Cruz. Using mice that express fluorescent proteins in cortical pyramidal neurons, we are able to image the process of synaptic change over time using repeated 2-photon imaging of cortical synapses (post-synaptic dendritic spines) through chronic cranial windows. Our application of this approach to understand post-stroke motor rehabilitation effects revealed that the training interacts with synapse turnover responses instigated by motor cortical lesions to created a new long lasting population of new synapses in remaining motor cortex (Fig. 7). An ongoing direction is to understand the interrelationships between neural and nonneuronal remodeling responses in the same region. We are also investigating the involvement of subventricular zone progenitor cells in these responses in collaboration with Dr. Michael Drew at UT.
Another newer direction is to understand how bimanual experiences influence patterns of brain reorganization after stroke like damage. Both hands are used together in most manual activities, yet the influence of bimanual experiences on brain reorganization has been neglected. We found that training rats after motor cortical infarcts in a novel cooperative bimanual skilled reaching task, the Popcorn Retrieval Task, improved unimanual performance with the impaired limb. We have a mouse version of this bimanual task in development to facilitate its combination with live animal imaging approaches.